Your surgical applications have specialized requirements.
Purpose-built AR meets them.

Adapted hardware.
Open software stack.
Regulatory-ready integration.
AR innovation that is industrialized, certified, and scalable.
ARspectra enables MedTech innovators to integrate purpose-built Augmented Reality (AR) into regulated Medical Devices. We provide customizable AR hardware platforms and open software architectures to be tailored to application-specific optical, tracking, mechanical, and regulatory requirements.
Through structured co-development, compliant manufacturing, and long-term supply assurance, we provide the technical and industrial foundation for scalable, AR-enabled medical products.
Align your medical AR platform with your application & procedure.
We engineer hardware platforms with medical specialties, procedures, and critical workflows in mind.
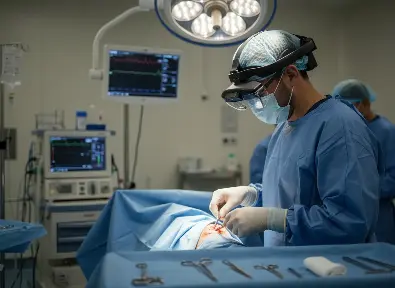
Wide Tracking area
Large sensor FOV
Focused tracking area
High precision inputs
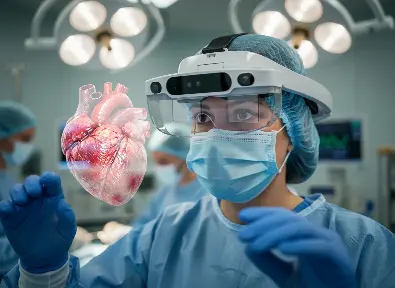
Fluorescence surgery
Optical fluorescence
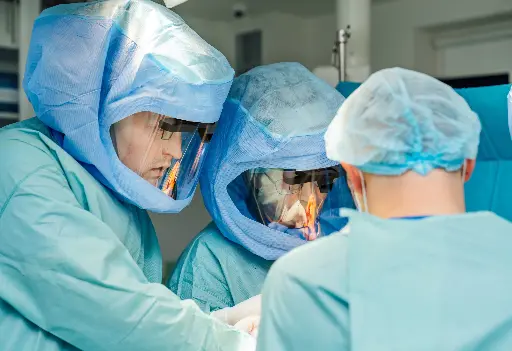
Orthopedic Surgery
Helmet integration

We adapt the hardware to the procedure - not the procedure to the hardware
1
Advanced AR hardware
powering our partners' next-generation Medical Devices.
Optical & Display Engineering
- High-brightness see-through AR displays
- 2D and stereoscopic 3D architectures
- Optimized for bright OR environments
Mechanical customization
- Fully customizable form factor
- Helmet-integrated designs
- Loupe-compatible configurations
- Lightweight, all-in-one
Tracking architecture
- Configurable camera specifications
- 3D TOF, SLAM, and others
- Adjustable optics for procedure-specific precision
Performance architecture
- High-performance processors
- Custom configurations
- Embedded optimization
2
Technical Excellence meets Clinical Reality.
Designed WITH & FOR the experts.
Procedure & usability adapted
Helmet-compatible designs
Integrated Surgical Headlights
Loupe integration

3
Open Software Architecture. Embedded-level support.
AR software platforms tailored to MedTech integration requirements.
Support from SDK to embedded architecture, for customization to specific medical applications.

Android-based systems
- Modification-ready Linux and RTOS compatibility.
- Unity, Unreal, Vulkan, and OpenGL support.
Open XR compatibility
- Standard AR development environments.
- Facilitates transfer from prior AR platforms.
Embedded optimization
- Performance fine-tuning.
- Direct sensor & interface access.
4
Certified. Regulatory-Aligned.

ARspectra operates as a fully certified MedTech manufacturer.
All devices, software and processes are aligned with our partners’ required medical-grade standards and certifications.
ISO 13485:2016
ISO 27001:2017
IEC 60601-1
EN ISO 14971
IEC 60601-1-2
IEC 62304
EN ISO 10993
MDSAP
And many more
5
Industrialized production. Assured supply. Long-term stability.
Developing a high-performance AR-based Medical Device is only part of the equation. Long-term production stability, traceability, and post-market support are critical.
Certified medical-grade manufacturing
Controlled software cycles
72-hour replacement programs
Adapted production & supply scaling
Global logistics support
Digital & field-based service
Why MedTech leaders choose ARspectra?
8+ years of multiple proprietary medical AR systems
Fully certified MedTech manufacturer
Customizable hardware architecture
Open software stack
Regulatory integration support
Long-term supply & service assurance
We provide the medical AR foundation on which next-generation Medical Devices are built.

Medical AR is not a trend.
It's a visualization & navigation shift!
Join us in building the future of medical AR!